
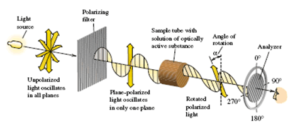
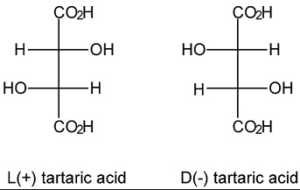
Naturale Racemico o Sintetico? Sino agli anni ‘90 il Tartrato Sodico Potassico veniva distinto in due tipi: Naturale e Sintetico. Con naturale si intendeva il prodotto che veniva ottenuto recuperando i tartrati prodotti dalla natura: essenzialmente derivati dall’ industria vinicola, questo prodotto è otticamente attivo (ruota di un angolo alfa un fascio di luce polarizzata) e denominato più correttamente come L+ Tartrato di Sodio e Potassio, è abbastanza tipico che i prodotti naturali, laddove vi sia la possibilità che essi siano otticamente attivi, siano disponibili esclusivamente in una sola delle forme otticamente attive. Con sintetico si intendeva un prodotto ottenuto per sintesi da reattivi ottenuti come derivati del industria petrolifera: reazione chimica tra anidride maleica e acqua ossigenata per ottenere acido racemico e a sua volta i sali racemici: Questo prodotto non è in grado di ruotare un fascio di luce polarizzata in quanto è una miscela al 50% di L+ Tartrato di Sodio e Potassio e 50% di D- Tartrato di Sodio e Potassio (dove l’ uno ruota la luce a destra, e l’altro la ruota a sinistra: ne consegue che essendo la miscela 50/50 la luce non ruota da nessuna parte) è tipico delle reazioni chimiche non stereoselettive, di portare alla formazione di racemi otticamente inattivi. I due componenti di questa miscela non sono separabili con metodi fisici. Ne consegue che i due prodotti sono facilmente distinguibili l’uno dall’altro. Dopo gli anni 90 alcuni produttori sono riusciti a sintetizzare per via chimica un prodotto assolutamente uguale a quello di origine naturale, ovvero otticamente attivo. Paradossalmente questo prodotto da un punto di vista chimico viene chiamato “naturale” per distinguerlo da quello racemico, anche se la sua origine è sintetica… Tutto ciò genera spesso confusione tra i non addetti ai lavori, e a volte anche tra gli addetti ai lavori meno esperti. Come si può distinguere il prodotto naturale “vero” da quello di origine sintetica? A parole oggi si parla di “prodotto naturale di origine vinica” per mettere in evidenza che esso è stato messo a disposizione direttamente dallal natura nel ciclo di produzione del vino e di “prodotto naturale ottenuto per sintesi” per sottolineare che quest’ultimo pur chiamandosi naturale (in quanto otticamente attivo) è comunque un prodotto di origine sintetica. Da un punto di vista analitico non vi sono a tutt’oggi metodi pratici riconosciuti per distinguere il prodotto naturale di origine vinica da quello di origine sintetica se non il metodo al Carbonio 14, un metodo estremamamente costoso. Per l’utilizzatore finale che cosa cambia? All’apparenza non cambia nulla, però, qualche dubbio e qualche interrogativo è meglio porselo sin da subito specialmente se si tratta di uso alimentare (ma anche per altri usi tecnologici e meglio essere prudenti): I limiti di purezza che il legislatore ha fissato per il prodotto naturale sono derivati dai tipi di processo tradizionalmente usati per ottenerlo, sono pertanto mirati a tenere sotto controllo le impurità che possono contaminarlo in funzione del processo che lo ha generato; la logica vorrebbe che per il prodotto sintetico si ricercassero i contaminanti tipici della sintesi (catalizzatori etc.) e non i contaminanti tipici del prodotto naturale cosa che invece oggi accade… Il risultato è che il prodotto naturale-sintetico all’apparenza sembra essere più puro del prodotto naturale, ma questo avviene solo perchè si vanno ricercare le impurezze tipiche del prodotto naturale vinico, mentre non si cercano le impurezze tipiche del prodotto ottenuto per sintesi, anzi non si sa neppure quali e quante esse possano essere, e ciò è assolutamente inquietante, specialmente se si pensa ad un utilizzo alimentare ed a eventuali effetti sul consumatore che potrebbero presentarsi anche a medio e lungo termine.
History
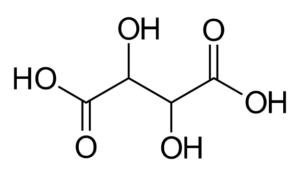
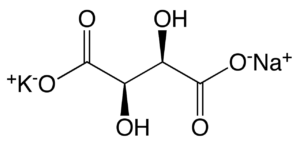
Il tartrato di sodio e potassio è il sale sodico potassico dell’acido tartarico. ACIDO TARTARICO L’acido tartarico storicamente ha una notevole importanza poichè sin dagli albori della chimica (era stato isolato gia nell’800 dopo cristo dall’alchimista Jābir ibn Hayyān) era noto come uno dei pochi acidi disponibili anche allo stato solido, con tutti i vantaggi pratici che al tempo ciò implicava. Il Tartrato di Sodio e Potassio fu preparato per la prima volta, attorno al 1675, dal farmacista francese Pierre Seignette, in onore del quale è noto con uno dei suoi sinonimi :”Sale di Seignette”; ma questo sale è anche noto (specialmente nel mondo anglosassone) con un altro sinonimo, ovvero Sale di Rochelle, in onore della cittadina francese di La Rochelle, in cui il dottor Seignette lo produsse per la prima volta. TARTRATO SODICO POTASSICO Nel 1847 Luis Pasteur, continuando gli studi di Jean Baptiste Biot, sulla capacità di questo sale di ruotare un fascio di luce polarizzata, riusci a separare manualmente ,cristallo per cristallo, una miscela racemica di questa sostanza: fu la prima volta che venne separata una miscela racemica di due enantiomeri (detta Racemo).
Uses
Tra gli usi più conosciuti del Tartrato Sodico Potassico ricordiamo i seguenti: • Come additivo nell’ industria alimentare: E337 utilizzo come conservante. • Nell’industria delle pectine. • Nell’industria delle gelatine alimentari • Per la preparazione della carta di sigarette come regolatore della combustione. • Nell’ industria degli specchi. • Nell’ industria galvanica. • Nell’industria del gesso e dei cementi come regolatore dei tempi di presa e per il controllo del volume finale dei manufatti. • Nella preparazione del reattivo di Fehling. • Nella preparazione del reattivo di Biuret. • Nell’industria elettronica. • Nell’industria delle paste dentifrice.
Packaging
Table 01 (title)
- Sacco di carta da 25 Kg
- Fusti in Fibra da 50 Kg
- Bancale di sacchi di carta da 1000 Kg
- Bancale di fusti in fibra da 900 Kg
Table 02 (title)
- Sacco di carta da 25 Kg
- Fusti in Fibra da 50 Kg
- Bancale di sacchi di carta da 1000 Kg
- Bancale di fusti in fibra da 900 Kg
Certificazione
CERTIFICATO FSSC 22000CERTIFICATO KOSHERCERTIFICATO HALAL
Specifiche
Scheda Tecnica SALE DI SEIGNETTE POLVEREScheda Tecnica SALE DI SEIGNETTE GRANULARE